The Trustwell Summer 2023 release for FoodLogiQ products brings a navigation makeover, enhanced insights, and new dashboard documentation widgets.
The development team at Trustwell has been hard at work this summer creating a variety of enhancements and new features to our FoodLogiQ products. In the spring, we announced new product names, greater flexibility in reviewing items, and updates in aiding your FSMA 204 compliance.
This time, Trustwell has continued our commitment to innovation with an updated look and feel, clearer data visualization, product attribute enhancements, and much more. We’re excited to share these product updates with our customers, so grab your shades along with a refreshing beverage, and let’s dive into how we’ve reshaped the FoodLogiQ platform this summer.
Revamped Look and Feel, Plus Enhanced Data
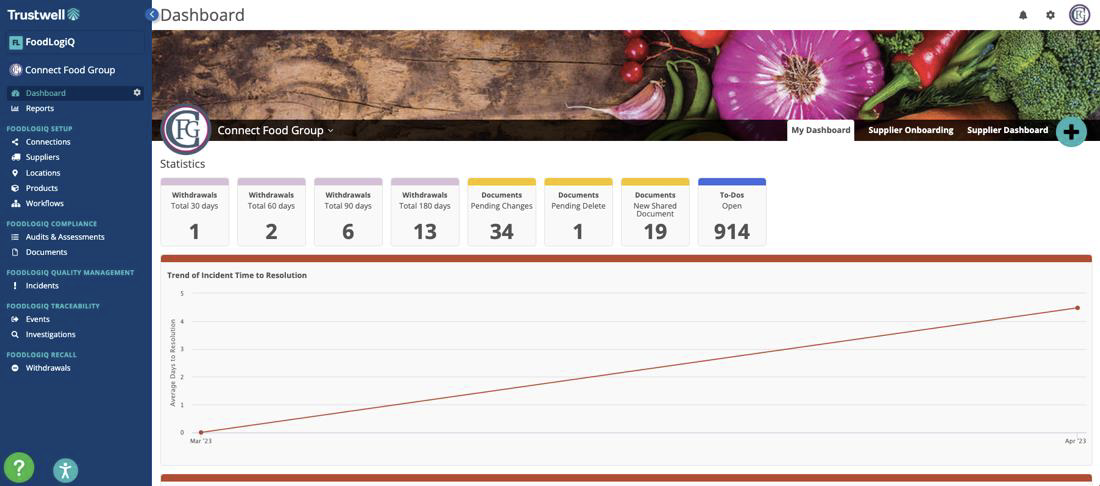
FoodLogiQ has undergone a makeover to embrace Trustwell's branded colors. Users will immediately notice a vibrant blue transformation as our formerly green action buttons now match our new aesthetic. Additionally, we’ve made the left navigation menu collapsible to provide you with more space on the screen without compromising functionality.
We understand the importance of clear data visualization. That's why we've reimagined the layout of large dashboard widget bar graphs. Data is now presented from greatest to least value for easy comprehension and quick analysis so your teams can make informed decisions.
GS1 Global Data Model and FSMA 204 Alignment
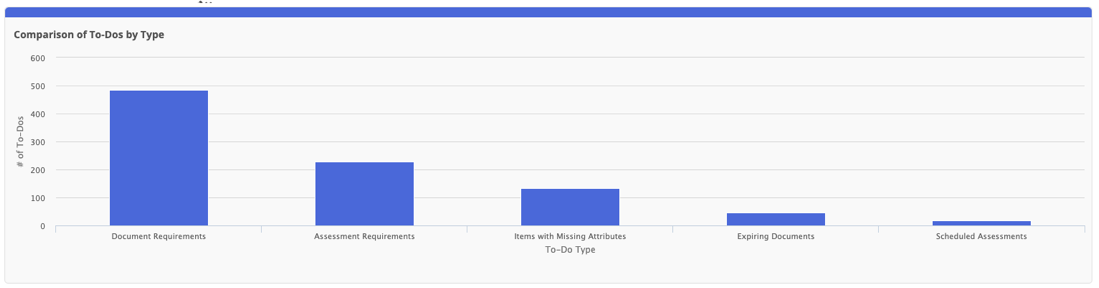
In alignment with the GS1 Global Data Model and FSMA 204 regulations, we’ve expanded our product attribute options. Users will be able to configure the new Packaging Type attribute for easy data entry and to create informative import/export templates.
Product Attribute, Gross Weight, Net Contents, and Utilization
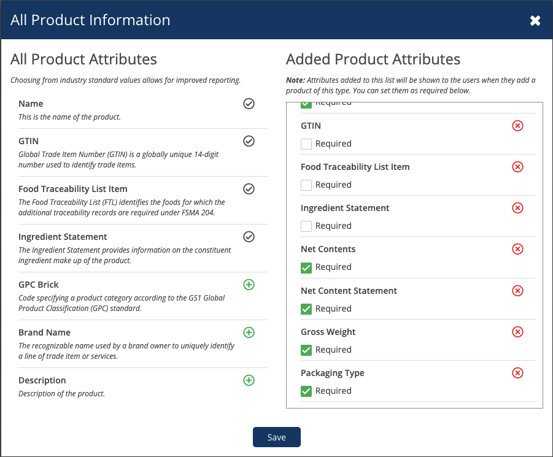
To add to the data enhancements, we’ve added a new attribute for Gross Weight and a numeric attribute for Net Contents. Now, you can input the numerical weight and corresponding unit of measure of your products for more accurate product information.
Lastly, users can now utilize the Food Traceability List and Packaging Type attributes while selecting document or audit requirements based on specific attribute responses. This flexibility ensures tailored and effective compliance management.
Streamlined Document Reviews and Collection
Three new dashboard widgets for documents have been added to offer valuable insights to FoodLogiQ customers. Quickly visualize new and changed documents by selecting the corresponding widget that will bring you to a filtered view of your documents. This simplified process makes document management more intuitive than ever.
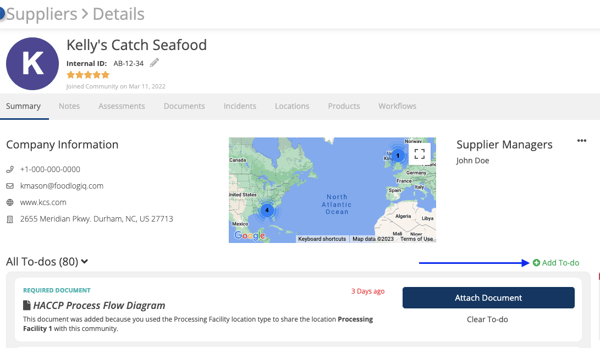
FoodLogiQ customers can also now request ad-hoc documents from their suppliers. Users can generate a to-do directly from the supplier information page, in order to request a document on a flexible, case-by-case basis. This enhancement quickens the document collection process and offers smoother collaboration with suppliers.
Recall Widgets for Enhanced Visibility
To further empower you in swift recall management, we’ve introduced four small dashboard widgets for recall management. These widgets display a count of open or closed withdrawals over 30-, 60-, 90-, or 180-day periods. Now, you can stay on top of all recall activities at a glance.
Read the Full Release Notes and More
Beyond the enhancements listed above, we’ve also made the following minor improvements:
- Users will now see the addition of Wiltshire to the list of available regions for the United Kingdom within our Locations feature.
- Reviewers assigned to workflow steps for your suppliers can now mark the step as complete by simply clicking the Complete button.
- Users who are configuring an email notification within an incident step will now be able to send a preview to any desired email address in real-time.
For more information on this release, check out these FoodLogiQ Knowledge Base articles related to this release:
For further information on the technical updates for this release, reach out to Release@trustwell.com for questions.
Tag(s):
Food Industry
,
Trustwell News
,
Traceability
,
Trustwell Software
,
FoodLogiQ News
,
Supplier Management
Other posts you might be interested in
View All Posts
Trustwell News
6 min read
| February 15, 2023
ESHA Research and FoodLogiQ Merge to Form Trustwell
Read More
Trustwell News
4 min read
| November 16, 2023
Fall 2023 FoodLogiQ Release: Enhanced Data Presentation, Recall Management, Streamlined Incident Reporting
Read More
Trustwell News
5 min read
| August 23, 2024

